MISSION IN MULTIPLE MYELOMA_
Could ongoing research on novel binding domains help build the future of BCMA therapies?1,2
Imagery is for illustrative purposes only and does not represent any specific binder.

The ongoing exploration of CAR binding domains has shown that differences in structure and related features may have an effect on CAR function4-8
CAR T-cell therapy has revolutionized MM treatment, but there is still a need for optimization.9
Potential for improved performance
Multiple myeloma, currently considered to be an incurable disease, accounts for about 10% of hematological malignancy diagnoses.9,10
Despite advances in MM treatment, including CAR T therapy, most patients eventually relapse.10,11
Reasons for relapse may include incomplete elimination of MM cells, as well as T-cell exhaustion.10,12
Further research is needed to address the unmet need in a broad range of patients with MM, including in high-risk subgroups.13
Safety considerations
CRS, ICANS, neurological side effects, hematologic toxicity, and infections are often associated with CAR T therapy.14
Logistical challenges
Variable turnaround times, manufacturing success rates, and slot availability may impact the accessibility of CAR T for patients with MM.14
While advancements have been made, further exploration into optimizing CAR T function is needed.9
Exploration of alternative binding domains for BCMA-directed CAR T-cell therapy in MM is underway1
Recent findings show that differences in binding domain structure and related features may have an effect on CAR function.4,5 There are multiple CAR binding domains being explored in pre-clinical studies, incorporating binding domains of various shapes and sizes.1
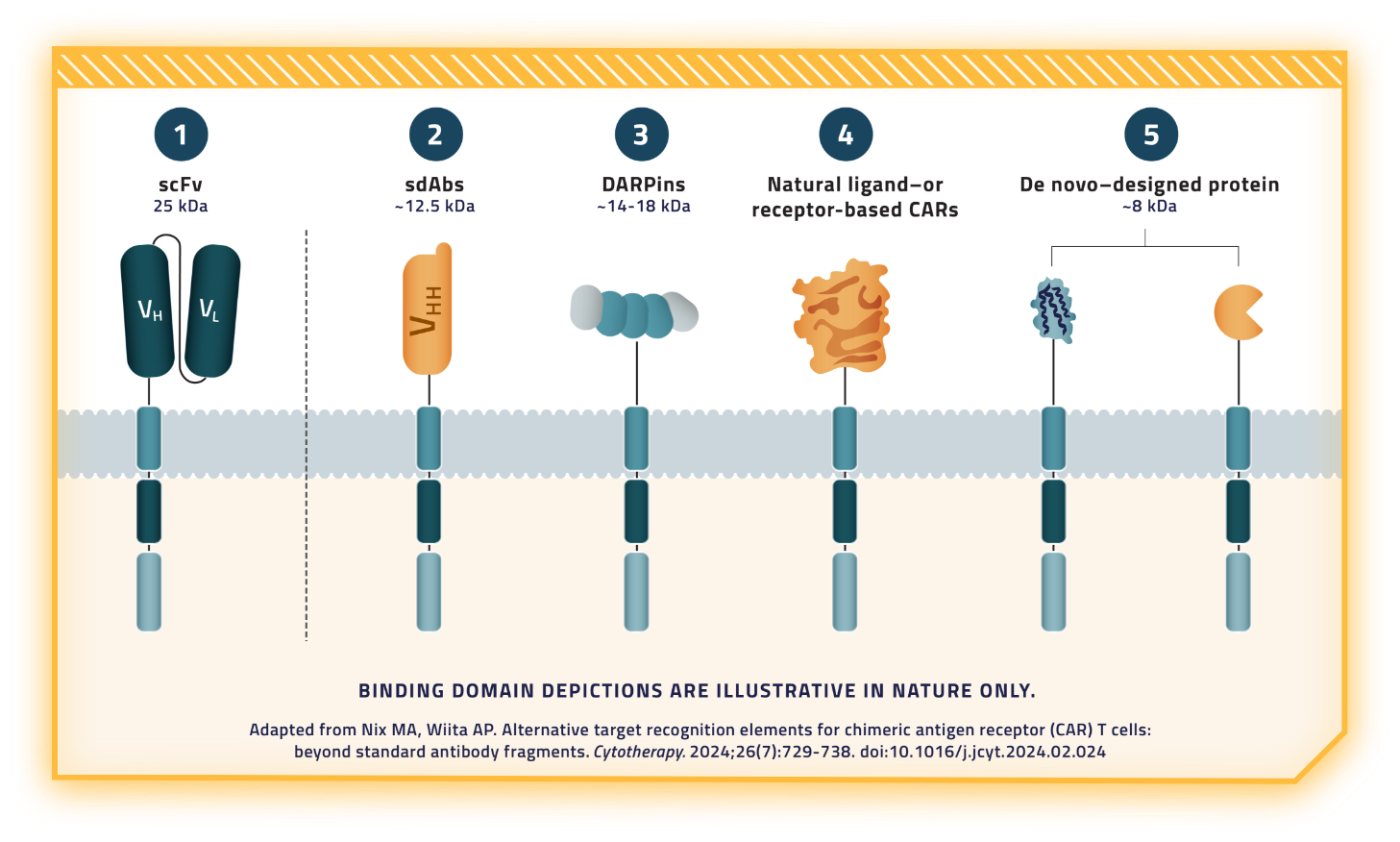
Select CAR binding domains include1,15:
- Single-chain variable fragments (scFv)—the most commonly used CAR, consists of variable regions of a heavy and a light chain
- Nanobodies or single domain antibodies (sdAbs)—variable heavy chain-only antibodies
- Designed ankyrin repeat proteins (DARPins)—multiple, stacked repeats of partially synthetic ankyrin protein domains
- Natural ligand– or receptor-based CARs—uses natural pairings of protein receptor-ligand interactions
- De novo-designed protein—fully synthetic protein binding elements (D-Domain, Co-LOCKR proteins)
Additional CARs (not shown) include adnectins, thermo-stable DNA binding proteins, and affibodies.16-18
The information provided is based on current understanding and may change in the future.
CAR design could matter in the development of the future treatment landscape4,5
Research in CAR T continues to evolve. The untapped potential in CAR design parameters, such as the binding domain, is being further investigated in pre-clinical studies.1,4,5,16,19-21
Based on pre-clinical data. Correlation of pre-clinical data with clinical effect has not been established.
Per current understanding and may change in the future.
Certain properties of alternative CARs being explored—such as small size, high stability, and no need for additional protein folding and assembly—may evolve the treatment landscape.16,19,21
Exploration to uncover potential in binding domain innovation in CAR T treatment landscape
Could unique binding domains have the potential to yield innovation in the CAR T treatment landscape?
CAR=chimeric antigen receptor; co-LOCKR=colocalization-dependent latching orthogonal cage/key proteins; CRS=cytokine release syndrome; DARPins=designed ankyrin repeat proteins; DNA=deoxyribonucleic acid; EMD=extramedullary disease; ICANs=immune cell-associated neurotoxicity syndrome; MM=multiple myeloma; scFv=single-chain variable fragments; sdAbs=single domain antibodies.
References:
1. Nix MA, Wiita AP. Alternative target recognition elements for chimeric antigen receptor (CAR) T cells: beyond standard antibody fragments. Cytotherapy. 2024;26(7):729-738. doi:10.1016/j.jcyt.2024.02.024 2. Swan D, Murphy P, Glavey S, Quinn J. Bispecific antibodies in multiple myeloma: opportunities to enhance efficacy and improve safety. Cancers. 2023;15(6):1819. doi:10.3390/cancers15061819 3. Sterner RC, Sterner RM. CAR-T cell therapy: current limitations and potential strategies. Blood Cancer J. 2021;11(4):1-11. doi:10.1038/s41408-021-00459-7 4. Burns WR, Zhao Y, Frankel TL, et al. A high molecular weight melanoma-associated antigen‐specific chimeric antigen receptor redirects lymphocytes to target human melanomas. Cancer Res. 2010;70(8):3027-3033. doi:10.1158/0008-5472.can-09-2824 5. Lakhani A, Chen X, Chen LC, et al. Extracellular domains of CARs reprogramme T cell metabolism without antigen stimulation. Nat Metab. 2024;6(6):1143-1160. doi:10.1038/s42255-024-01034-7 6. Ghorashian S, Kramer AM, Onuoha S, et al. Enhanced CAR T cell expansion and prolonged persistence in pediatric patients with ALL treated with a low-affinity CD19 CAR. Nat Med. 2019;25(9):1408-1414. doi:10.1038/s41591-019-0549-5 7. Drent E, Themeli M, Poels R, et al. A rational strategy for reducing on-target off-tumor effects of CD38-chimeric antigen receptors by affinity optimization. Mol Ther. 2017;25(8):1946-1958. doi:10.1016/j.ymthe.2017.04.024 8. Greenman R, Pizem Y, Haus-Cohen M, et al. Shaping functional avidity of CAR T cells: affinity, avidity, and antigen density that regulate response. Mol Cancer Ther. 2021;20(5):872-884. doi:10.1158/1535-7163.mct-19-1109 9. Sheykhhasan M, Ahmadieh-Yazdi A, Vicidomini R, et al. CAR T therapies in multiple myeloma: unleashing the future. Cancer Gene Ther. 2024;31(5):667-686. doi:10.1038/s41417-024-00750-2 10. Zhang X, Zhang H, Lan H, Wu J, Xiao Y. CAR-T cell therapy in multiple myeloma: current limitations and potential strategies. Front Immunol. 2023;14. doi:10.3389/fimmu.2023.1101495 11. Kumar S, Baizer L, Callander NS, et al. Gaps and opportunities in the treatment of relapsed-refractory multiple myeloma: consensus recommendations of the NCI Multiple Myeloma Steering Committee. Blood Cancer J. 2022;12(6):1-11. doi:10.1038/s41408-022-00695-5 12. Medina-Herrera A, María Eugenia Sarasquete, Jiménez C, Puig N, Ramón García-Sanz. Minimal residual disease in multiple myeloma: past, present, and future. Cancers. 2023;15(14):3687. doi 10.3390/cancers15143687 13. Dima D, Abdallah AO, Davis JA, et al. Impact of extraosseous extramedullary disease on outcomes of patients with relapsed-refractory multiple myeloma receiving standard-of-care chimeric antigen receptor T-cell therapy. Blood Cancer J. 2024;14(1). doi:10.1038/s41408-024-01068-w 14. Rendo MJ, Joseph JJ, Phan LM, DeStefano CB. CAR T-cell therapy for patients with multiple myeloma: current evidence and challenges. Blood Lymphat Cancer. 2022;12:119-136. doi:10.2147/blctt.s327016 14 15. Smith R. Bringing cell therapy to tumors: considerations for optimal CAR binder design. Antib Ther. 2023;6(4):225-239. doi:10.1093/abt/tbad019 16. Han X, Cinay GE, Zhao Y, Guo Y, Zhang X, Wang P. Adnectin-based design of chimeric antigen receptor for T cell engineering. Mol Ther. 2017;25(11):2466-2476. doi:10.1016/j.ymthe.2017.07.009 17. Zajc CU, Dobersberger M, Schaffner I, et al. A conformation-specific ON‐switch for controlling CAR T cells with an orally available drug. Proc Natl Acad Sci USA. 2020;117(26):14926-14935. doi:10.1073/pnas.1911154117 18. Salzer B, Schueller CM, Zajc CU, et al. Engineering AvidCARs for combinatorial antigen recognition and reversible control of CAR function. Nat Commun. 2020;11(1):4166. doi:10.1038/s41467-020-17970-3 19. Arbabi-Ghahroudi M. Camelid Single-domain antibodies: historical perspective and future outlook. Front Immunol. 2017;8. doi:10.3389/fimmu.2017.01589 20. Long AH, Haso WM, Shern JF, et al. 4-1BB costimulation ameliorates T cell exhaustion induced by tonic signaling of chimeric antigen receptors. Nat Med. 2015;21(6):581-590. doi:10.1038/nm.3838 21. Siegler E, Li S, Kim YJ, Wang P. Designed ankyrin repeat proteins as Her2 targeting domains in chimeric antigen receptor-engineered T cells. Hum Gene Ther. 2017;28(9):726-736. doi:10.1089/hum.2017.021 22. Fujiwara K, Masutani M, Tachibana M, Okada N. Impact of scFv structure in chimeric antigen receptor on receptor expression efficiency and antigen recognition properties. Biochem Biophys Res Commun. 2020;527(2):350-357. doi:10.1016/j.bbrc.2020.03.071 23. Jayaraman J, Mellody MP, Hou AJ, et al. CAR-T design: elements and their synergistic function. EBioMedicine. 2020;58:102931. doi:10.1016/j.ebiom.2020.102931 24. Mazinani M, Rahbarizadeh F. CAR-T cell potency: from structural elements to vector backbone components. Biomark Res. 2022;10(1):70. doi:10.1186/s40364-022-00417-w 25. Roddie C, Dias J, O’Reilly MA, et al. Durable responses and low toxicity after fast off-rate CD19 chimeric antigen receptor-T therapy in adults with relapsed or refractory B-Cell Acute lymphoblastic leukemia. Journal of Clinical Oncology. 2021;39(30):3352-3363. doi:10.1200/jco.21.00917. 26. Hiratsuka H, Yasushi Akahori, Shingo Maeta, Egashira Y, Hiroshi Shiku. Fast on-rates of chimeric antigen receptors enhance the sensitivity to peptide MHC via antigen rebinding. Journal of Biological Chemistry. 2024;300(9):107651-107651. doi:10.1016/j.jbc.2024.107651.